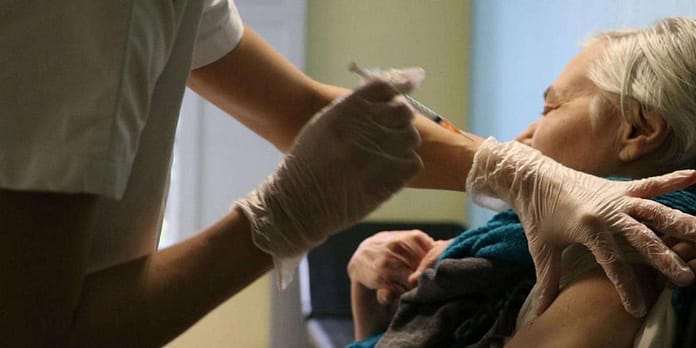
The flu is coming to the northern hemisphere and with it the vaccination campaign. If, to date, vaccines against this virus use techniques that are known but are not 100% effective, the emergence of mRNA could change the rules of the game.
More and more laboratories are beginning to develop vaccines against the influenza virus using this new technology. And so Sanofi, the global leader in influenza, has begun trials of a monovalent RNA vaccine — targeting one strain of the virus — and will begin trials on a quadrivalent vaccine next year.
US company Pfizer in September embarked on the first injection of an anti-flu vaccine into humans using messenger RNA, already used in its Covid-19 vaccine. US biotech company Moderna launched its own trials in early July.
What is the benefit of this technology that has been proven to be effective against Covid-19 but has never been used against other viruses?
“increase efficiency”
Flu vaccines have been around for a long time. However, its effectiveness is not complete: it uses inactivated viruses, which must be prepared well in advance, to achieve an efficacy of 40-60%, or even 70%. Six months before the outbreak of the epidemic, we evaluate the most prevalent strains. Sometimes we make mistakes, and that leads to a significant increase in mortality,” explains Claude-Agnes Raynaud, immunologist and director of research at Inserm.
Additionally, “the problem with virus inactivation in a vaccine preparation is that it can damage certain surface proteins,” she says, those that stimulate the immune response.
Conversely, messenger RNA does not need to produce antigens (a substance foreign to the body that triggers an immune response) in millions of eggs, because it will be produced by the human cell, and even virus proteins.
Jean-Jacques Le Four, an analyst at Bryan and Garnier & Co. With the switch, the efficiency increased which can reach 95%. So many researchers are on the right track.
cons
However, this technology has drawbacks, including extremely low temperature storage conditions.
“We will have to access heat-resistant vaccines, which can be stored in the refrigerator at 2 to 8 degrees, in syringes. There are many things that need to be done to convert messenger RNA into influenza,” explained recently Thomas Triumph, Vice President of Vaccines at Sanofi.
Without forgetting “the question of acceptability: by the time these vaccines arrive, will the population be confident about this technology or will they always be hesitant?” ‘ asks Jean-Jacques Le Four. However, it is not enough to suppress appetite. “Sanofi has realized that it cannot ignore this technology. Influenza vaccines represent 2.5 billion euros in sales each year.”